Evaluating Research Quality Using Critical Appraisal Tools
What is critical appraisal?
Critical appraisal is the process of carefully and systematically assessing the strengths and limitations of a research study to determine its quality and suitability for informing clinical decision-making. The critical appraisal process involves evaluating a study’s design, conduct, results, and interpretation to evaluate its quality and usefulness. The types of questions asked during a critical appraisal vary based on the level of evidence.
Levels of Scientific Evidence
Determining the best evidence is guided by a hierarchical system that classifies evidence. This hierarchy is known as the levels of evidence. This hierarchy is a systematic process designed to rate evidence (research literature) when determining the effectiveness of a particular intervention. The hierarchies rank studies according to the probability of bias occurring in the research design or interpretation of the results. Among the primary sources of evidence (RCTs, cohort studies, case-controlled studies, case series, and reports), RCTs are given the highest level because they are designed to be unbiased and have less risk of systematic errors. A case series or expert opinion is often biased by the author’s experience or opinions and has limited control of confounding factors, therefore will be ranked as low on the hierarchy. Systematic reviews, including meta-analyses and meta-syntheses, are at top of the hierarchy, as these studies are used to develop clinical guidelines.
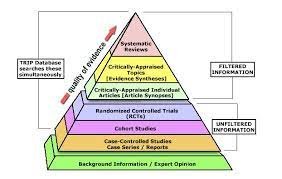
Components of a Critical Appraisal Tool
There are a number of different elements that can be included as part of a critical appraisal tool. The specific components will vary depending on the particular tool being used, but some common elements include:
- A description of the research study design. This should include information on how the study was conducted, what type of data was collected, and how the data was analyzed.
- A discussion of the research question(s) being addressed by the study. This should include an evaluation of whether the question(s) is/are clearly defined and relevant to the topic under investigation.
- An assessment of the quality of the data collected in the study. This should include an evaluation of whether the data is valid and reliable.
- A discussion of the study’s findings in relation to the research question(s). This should include evaluating whether the findings are clear and supported by the data presented in the study.
- An assessment of any limitations associated with the study design or analysis. This should include an evaluation of whether any potential sources of bias or error could have affected the study results.
Click here to view a published critical appraisal.
Types of Critical Appraisal Tools
Many different critical appraisal tools (CATs) are available, each with its own strengths and weaknesses. Choosing an appropriate CAT based on the research methodology and study design is important. For example, if you evaluate the quality of a randomized controlled trial (RCT), you must use a CAT for RCTs.
CATs are available for the following methodologies and research designs:
-
Randomized controlled trial (RCT)
-
Systematic reviews
-
Cohort studies
-
Diagnostic studies
-
Case control studies
-
Mixed methods studies
-
Qualitative studies
The following websites contain CATs for each of the above study types:
- Critical Appraisal Skills Programme: https://casp-uk.net/casp-tools-checklists/
- Joanna Brigs Institute tools: https://jbi.global/critical-appraisal-tools


